Contract Analysis
ISO:9001 and GMP
Certified Laboratory
Paralab GMP Lab unlocks quality through compliance. With a team of experienced scientists and cutting-edge technology, we relentlessly focus on innovation and precision. Our GMP-certified laboratory offers a comprehensive suite of services tailored to the pharmaceutical industry. We customize our services to meet your specific needs, whether it is supporting API development, conducting detailed characterization, method development, performing reverse engineering, or conducting root cause analysis. With our expertise and capabilities, we aim to be your trusted partner in achieving success in the competitive pharmaceutical landscape.
Serviço de Análises
Laboratório Certificado
ISO:9001 e GMP
Dispomos de um serviço de análises em várias técnicas instrumentais, destinado às várias áreas: desde a área dos materiais até à área das biociências. Investimos nas infraestruturas e na qualidade dos nossos recursos humanos de forma a assegurar o melhor serviço.
Destacamo-nos pela experiência dos nossos especialistas nas diversas técnicas e respetivas áreas de aplicação. A nossa missão é satisfazer as suas necessidades tanto na realização de análises, desenvolvimento e validação de métodos; como na investigação de falhas ou causa raiz nas investigações de engenharia reversa.
SUPPORT & RESEARCH
Let's explore your sample together!
We distinguish ourselves by support we provide to our clients and by our research into differentiating parameters of product and/or process quality. A report is issued for each analysis request in Spanish, English or Portuguese.
Contact us
Our lab specialists are here to help you.
ANALYSIS/TECHNIQUES
Types of analyses
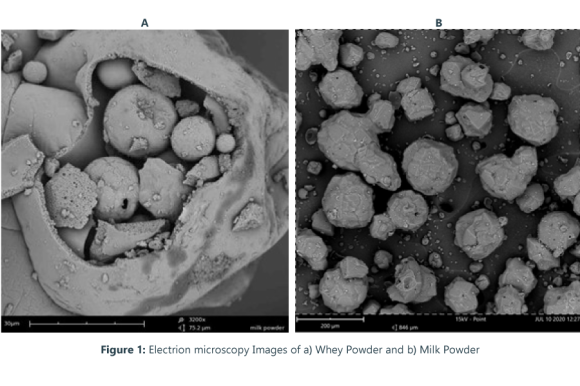
Scanning Electron Microscopy (SEM) is a technique applied to the characterization of materials. This technique allows the observation and characterization of the morphology of a sample, showing surface details up to the nanometric scale.
With this technique, we can observe the detailed morphology of the surface, as well as perform a dimensional analysis of the sample, measure its roughness, particle size, and determine the preferred location of elements (elemental mapping), among others.
Our benchtop SEM-EDS is equipped with:
Backscatter Electron Detector (BSED), which provides higher image contrast (sample areas where elements with higher atomic number are present appear lighter in the image) and enables the analysis of non-conductive samples without coating;
Secondary electron detector (SED) that provides more detailed surface and topographic information by detecting electrons diffused inelastically by the material;
Detector EDS (Energy Dispersive Spectroscopy) provides information on elemental composition of a sample.
Maximum magnification: 150000x
Resolution up to 17 nm (sample dependent)
Application Examples
Laser Diffraction is an experimental analysis technique that allows determining sizes and size distribution of particles in a range of a few nanometres to a few millimetres. A quantitative size distribution of the entire population of particles is obtained to determine which diameters represent a certain percentage of that population. The distribution can be in number or, more usually, in volume.
The theoretical models that allow the calculation of particle diameters are Fraunhofer’s and Mie’s. According to ISO 13320-1, Fraunhofer theory gives good results for particle sizes above 50 microns. For smaller sizes, in general, Mie’s theory is more appropriate. Mie’s theory is more complete and correct because it considers a certain degree of transparency of the particle to the laser, however this implies prior knowledge of the optical properties of the sample, namely its Refractive Index (RI) and Absorption Index (AI). Laser diffraction has proven useful for the following industries: environmental, ceramics, pharmaceutical, food and cosmetics.
The determination of particle size and distribution by the laser diffraction technique is recognized by numerous standards and standardization agencies including: ISO, ASTM, and USP.
Application Examples
Dynamic Light Scattering (DLS) is a technique that is commonly used to measure particle size and can estimate the distribution of submicrometere particles in scattering. DLS analyzes the hydrodynamic mobility of the particles. The success of the technique is mainly based on the fact that it provides estimates of the average particle size and size distribution within a few minutes.
Electrophoretic light scattering (ELS) is an indirect analysis of measuring electrophoretic mobility through the Doppler shift observed in light scattered by particles. In an ELS experiment, a beam of coherent light is focused on particles dispersed in a liquid and subjected to an electric field. The charged particles move toward either the anode or the cathode, depending on their charge on the medium. ELS provides fast, accurate, automatic, and highly reproducible electropherograms of complex particles dispersed in aqueous or non-aqueous media, without the need to use standard particles for calibration.
The mathematical models for correlogram analysis are the cumulant method for size and polydispersity; and the non-negative least squares method for size distribution. The Zeta potential is derived from the electrophoretic mobility using the Henry function, which can be approximated by the Smoluchowski equation or Hückel equation according to the relative thickness of the electrical double layer.
Application Examples
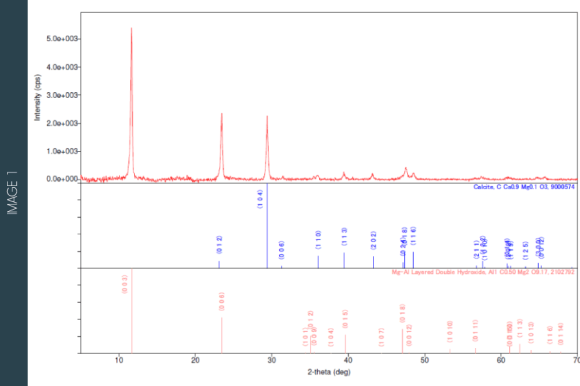
X-Ray Diffraction is a very versatile technique with broad application in the characterization of materials, since it allows the identification and quantification of different structural phases, determine crystallinity, and crystalline network parameters, gauge residual mechanical stress, among many other properties of powdered or whole materials, solids or liquids.
In the analysis of solid samples in refraction mode, the sample is irradiated by an X-ray beam which is subsequently refracted at certain angles according to the crystalline structure, according to Bragg’s Law of Refraction: nλ = 2d sinθ where n is the order of refraction (n = 1, 2, …), λ is the wavelength of the incident beam, d is the spacing between atomic planes in the crystal, and θ is the angle formed between the incident beam and the plane of refraction.
Since each substance has a unique spectrum, the relative number, and intensity of the peaks in a sample allow the identification of different phases present and therefore the determination of their qualitative composition. The quantitative composition can be determined using a calibration curve. Alternatively, it can be calculated using the RIR (Reference Intensity Ratio) method, where a pre-established proportionality factor is used between the most intense reflections of each phase and a standard substance (Al2O3-corundum).
Application Examples
Rheology is the study of the flow and deformation of materials when a force is applied, usually using a rheometer. Rheological properties are referred to all fluid materials, from dilute solutions of polymers and surfactants through concentrated protein formulations; semi-solid materials such as pastes and butter to, molten materials, and solid materials such as polymers and asphalt.
Many materials and formulations exhibit complex rheological properties, whose viscosity and viscoelasticity differ depending on the external conditions applied, such as stress, pressure, time, and temperature. Internal sample differences, such as chemical nature, concentration and stability, and the type of formulation are also key factors for rheological properties.
The type of rheometer required for measuring these properties often depends on the appropriate shear rates and time frames, as well as the sample size and viscosity.
Application Examples
Gas chromatography (GC) is an essential analytical technique in chemistry, providing valuable insights into substance composition and purity. It plays a central role across various industries, including pharmaceuticals. GC separates and quantifies volatile compounds in complex mixtures, offering precise results. In pharmaceuticals, GC ensures product quality and safety by identifying the purity of APIs, impurities present, degradation products, and residuals solvents. GC analysis is crucial for quality control and is recognized by numerous standards and standardization agencies, including ISO, ASTM, and USP.
Application Examples
The dissolution test is a fundamental tool in pharmaceutical analysis, offering valuable insights into the performance and consistency of drug products. It plays a vital role in ensuring the efficacy, safety, and quality of pharmaceutical formulations. This test provides critical information about the drug's release characteristics, which are essential for assessing its bioavailability and ensuring consistency in manufacturing. The dissolution test is described in the USP Chapter <711>.
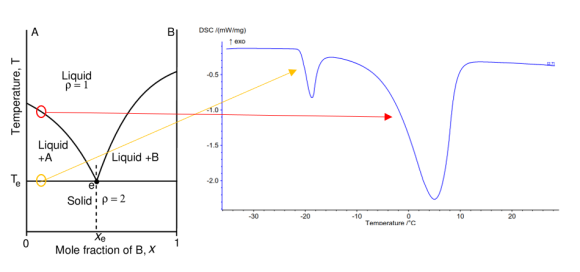
Differential Scanning Calorimetry (DSC) is one of the most widely used techniques in the thermal characterization of materials. In this technique, the heat flow between the sample and a reference is monitored when subjected to a controlled temperature program, which changes whenever there is a phenomenon in the sample that consumes or releases energy. These phenomena may be of a physical or chemical nature. Physical transformations include melting, crystallization, vaporization, and glass transitions, among others. On the other hand, chemical transformations involve reactions that include decomposition, combustion, chemical absorption, polymerization, solid-solid transitions, among others.
The DSC technique allows not only to determine the temperature at which a given endo/exothermal phenomenon occurs, but also to determine the amount of energy (enthalpy) involved in it. In addition to this type of information, the technique also allows one to measure heat capacity values of a material in accordance with ASTM E 1269, ISO 11357-4 and DIN 51007 standards, as well as determine degrees of purity or perform kinetic studies.
Application Examples
Thermogravimetry is a Thermal Analysis technique where the loss or gain in mass of a sample when subjected to a controlled temperature program is monitored. The changes in mass can be due to chemical processes, such as decomposition reactions with gas release or combustion reactions, and physical processes, such as vaporization of volatiles or absorption of moisture, among others.
The combination of this technique with others such as DSC or analysis of the gases released (FTIR, MS, GC-MS) allows a more concrete identification of the phenomena and allows inferences about the reaction mechanism. This fact, together with the mass loss associated with the phenomenon, enables the quantification of the different components that constitute the sample.
Application Examples
XRF
X-Ray Fluorescence (XRF) is a non-destructive analytical technique used to determine the elemental chemical composition of all types of materials except gases. For this reason it is the method of choice for many different types of applications, particularly in quality control of materials and industrial production. The XRF technique is excellent for both qualitative and quantitative analyses.
The technique is subdivided into two categories: Energy Dispersive XRF and Wavelength Dispersive XRF (ED-XRF and WD-XRF, respectively). The difference between the two lies in the signal processing and detection. In the ED-XRF technique, the X-ray photons emitted by the sample are detected simultaneously, usually by a solid-state semiconductor detector, while in WD-XRF the X-rays are diffracted by a crystal to be separated into their wavelengths and then detected. The ED-XRF technique is usually faster and the equipment is simpler and easier to use. WD-XRF equipment is usually larger, more complex and slower to measure, but can detect more elements (up to Beryllium), with better resolution and lower detection limits.
Application Examples
Quantification of Silicon in textile fibers
Verification of possible contaminations in flours resulting from milling
Elemental quantification of liquid paints
Elementary quantification of phosphoric rocks
Detection of defective auto parts
Quantification of chlorides remaining from the synthesis of new APIs
Liquid Density
The density of liquids is a specific property that can be used in the quality control of some products, such as milk and fuels.
Refractive Index
Determination of refractive index is important as it is a characteristic property of each substance and can therefore be used to identify them.
Elemental Analysis (DUMAS and CHNS)
Enables the determinination of quantitative composition of a material, identifying the elements present and their relative proportions. These analyses are commonly applied to samples of chemical substances, organic and inorganic compounds, metal alloys, minerals, among others.
ANALYSIS SERVICE TEAM
Multidisciplinary team
Our commitment is based on the team's long experience and multidisciplinary approach.
Bruno Machado, PhD
Materials & Chemistry
Hugo Gonçalves, PhD
Physics
José Catita, PharmaD, PhD
Pharmacy
Paulo Silva, PharmaD, PhD
Technical Director GMP
Regina Torre,
PhD student
Chemistry
Fábio Major,
MSc
Biological Chemistry
Knowledge
Application Notes
Porto
(Headquarter)
Rua Dr. Joaquim Manuel Costa, 946 B
4420-437 Valbom, Gondomar
Phone: +351 224 664 320*